Atom Air Incu I User Manual Download
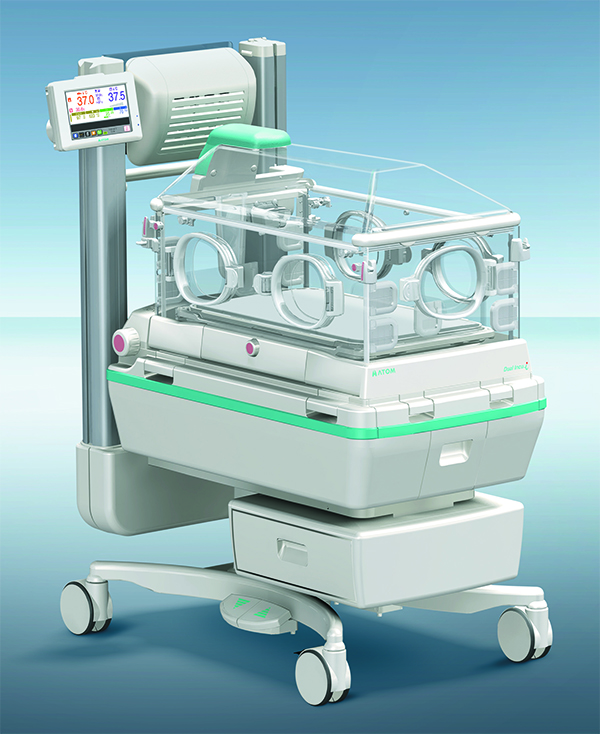
. AirMini setup pack - N20: Air tubing, N20 connector, HumidX ™ HumidX Plus OR. AirMini mask pack for P10: Air tubing, AirFit P10 for AirMini mask (M), P10 cushion (S), P10 cushion (L), HumidX, HumidX Plus, AirFit P10 headgear clips. Page 5 Your machine is compatible with ResMed's F20 and N20 series masks and with the AirFit P10 for. New generation of hybrid incubator evolved from incubator It is an high-end model in the Atom Infant Incubator series. It supplies two types of warming modes, a radiant heat source from the top, together with a convective heat source, when the hood is closed.
Manuals or user guides for your HP ProBook 4540s Notebook PC IBM WebSphere Portal. Home; IBM Logo. HP ProBook 4540s Notebook PC User Guides. HP Customer Support Search. Select your model Let HP find my products Identify now. HP can identify most HP products and recommend possible solutions. Hp probook 4540s user manual pdf. Hp ProBook 4540s Manuals Manuals and User Guides for HP ProBook 4540s. We have 6 HP ProBook 4540s manuals available for free PDF download: Maintenance And Service Manual, User Manual, Getting Started Manual, Quickspecs, Specification, Technical White Paper.
-
|
Atom Air Incu I User Manual 10
| |||||||||||||||||||||||||||
| Device Design | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Atom Medical sent an Urgent Field Safety Notice dated May 1, 2012, to all affected customers. The letter identified the product, the problem, and the action to be taken by the customer. Customers were instructed to immediately identify all affected product in their inventory and quarantine them until the rework was complete. Customers were also asked to identify any customers to whom they further distributed the product and to contact their customers at once and arrange a time in whcih the filed service technician could perform the rework. Customers were asked to return the Recall Response Forms to Atom Medical ([email protected], [email protected], or [email protected]). All forms should be completed and returned by January 31, 2013. For questions regarding this recall call 512-327-9997. | |||||||||||||||||||||||||||
| 377 units | |||||||||||||||||||||||||||
| Worldwide Distribution - USA including PA. Internationally to Japan, Kuwait, Saudi Arabia, Lenanon, Italy, Spain, China, East Europe, Slovakia, Poland, Czech Republic, Ukraine, Egypt, Russia, Algeria, Colombia, Germany, Malaysia, Switzerland, Australia, Europe, Portugal, Iran, Israel, Philippines, UAE, Korea, and Netherlands | |||||||||||||||||||||||||||
| TPLC Device Report | |||||||||||||||||||||||||||
| 1 A record in this database is created when a firm initiates a correction or removal action. The record is updated if the FDA identifies a violation and classifies the action as a recall, and it is updated for a final time when the recall is terminated. Learn more about medical device recalls. 2 Per FDA policy, recall cause determinations are subject to modification up to the point of termination of the recall. 3 For details about termination of a recall see Code of Federal Regulations (CFR) Title 21 §7.55. | |||||||||||||||||||||||||||
| 510(K) Database | 510(K)s with Product Code = FMZ and Original Applicant = ATOM MEDICAL CORPORATION | ||||||||||||||||||||||||||
